Imagine scientists deciding to solve a problem that kills over 600,000 people annually — by genetically modifying the mosquitoes themselves. It sounds like science fiction, but labs in London, Florida, and Africa are already testing this approach. The results are simultaneously promising and deeply unsettling.
📖 Read more: Pet Cloning: Same DNA, Different Soul
🦟 Why Target Mosquitoes
Mosquitoes aren't just annoying. They're the deadliest animal on the planet. Every year, female mosquitoes — the only ones that bite — transmit diseases like malaria, dengue fever, Zika virus, and chikungunya. Malaria alone killed 627,000 people in 2021, according to the World Health Organization. Most were children under five in sub-Saharan Africa.
Traditional methods — bed nets, insecticides, draining standing water — have helped, but progress has stalled. Since 2015, malaria cases have remained steady around 230 million annually. Worse, mosquitoes are developing resistance to insecticides and parasites are becoming resistant to drugs. Something radically different is needed.
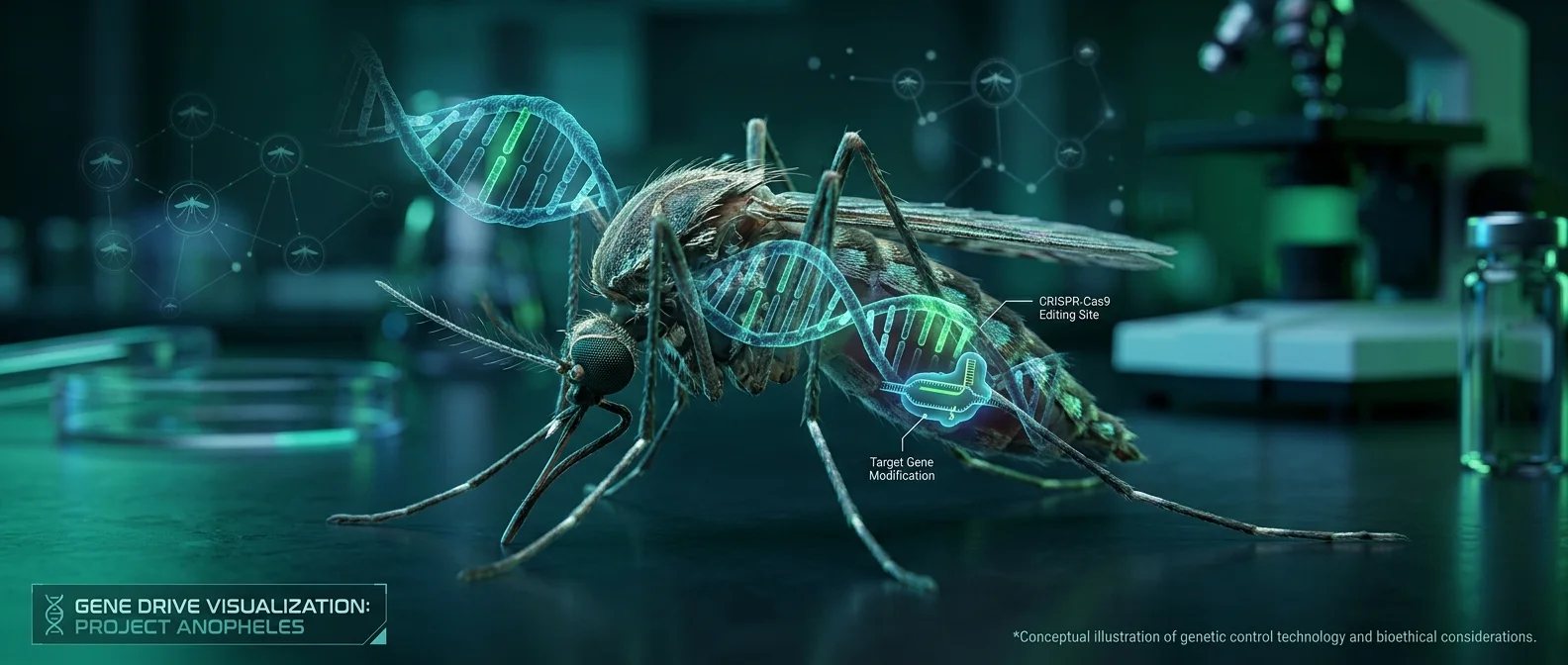
🔬 CRISPR and Gene Drives: The Tools
CRISPR-Cas9 technology — the “molecular scissors” — allows scientists to cut and modify DNA with surgical precision. In mosquitoes, it's used to introduce genes that prevent disease transmission or reduce their populations.
Gene drives are the most audacious application. Normally, a gene is inherited by 50% of offspring. A gene drive “breaks” this rule: it copies itself into nearly every offspring, spreading the modification through entire populations within a few generations. In the lab, this means releasing a small number of modified mosquitoes could theoretically alter an entire species.
There are two basic strategies: population suppression — reducing or eliminating the target species — and population replacement, where mosquitoes survive but lose their ability to transmit diseases.

🧪 Peptides That Kill the Parasite
A team at Imperial College London — Transmission:Zero — pursued a particularly elegant approach. Instead of killing mosquitoes, they modified them to produce antimicrobial peptides in their guts every time they feed on blood.
These peptides — originally isolated from bees and African clawed frogs (Xenopus laevis) — interfere with the energy metabolism of the Plasmodium parasite. The result? Parasite development is delayed by several days. This sounds trivial, but it's anything but.
Only about 10% of mosquitoes live long enough for the parasite to mature and reach the salivary glands. With the additional delay, virtually no mosquito survives long enough to become infectious before dying naturally. The modification also slightly reduces mosquito lifespan — a “double hit.” A 2022 study in Science Advances, led by Dr. Tibebu Habtewold and Astrid Hoermann, showed dramatic transmission reduction under laboratory conditions.
🧬 Population Collapse in One Year
The same Imperial team, collaborating with Polo GGB in Italy and the Liverpool School of Tropical Medicine, went a step further. They created a gene drive targeting the doublesex gene of Anopheles gambiae — the mosquito responsible for most malaria cases in sub-Saharan Africa.
The modified mosquitoes carry a mutation that destroys female fertility. The gene drive ensures this mutation is inherited almost invariably. In large-scale experiments — cages simulating natural conditions with controlled temperature, humidity, and sunrise/sunset simulation — the population collapsed completely within one year.
A striking finding: no resistance mutations emerged. This is crucial. Many feared natural selection would “break” the gene drive, but choosing the doublesex gene — an extremely evolutionarily conserved gene — appears to prevent this escape. The results were published in 2021 in Nature Communications by Dr. Andrew Hammond and colleagues.
🏭 Oxitec: First Mosquitoes in the Wild
While gene drives remain in laboratory stages, biotechnology company Oxitec has already released genetically modified mosquitoes in real cities. Their approach is more conservative: male Aedes aegypti carrying a “self-destruct” gene.
📖 Read more: CRISPR 2026: Can We Design Babies?
These males mate with wild females, but their offspring die before reaching adulthood. There's no gene drive — the modification disappears on its own, requiring continuous releases. In Brazil, trials in the city of Piracicaba recorded a 90% reduction in local larvae of the species. In May 2021, Oxitec began the first releases in the Florida Keys, despite strong opposition from residents.
The method primarily targets dengue fever, Zika, and chikungunya — diseases transmitted by Aedes aegypti. In contrast, gene drive technology focuses on Anopheles gambiae and malaria. Two different enemies, two different strategies.

🦠 Wolbachia: The Bacterial Alternative
"DNA scissors" aren't always necessary. The bacterial approach using Wolbachia represents an alternative being tested in dozens of countries. Wolbachia pipientis — an intracellular bacterium that naturally infects many insects but not Aedes aegypti — is artificially introduced into mosquitoes.
Infected mosquitoes struggle to transmit dengue, Zika, and chikungunya viruses. Simultaneously, Wolbachia spreads through the population thanks to a phenomenon known as "cytoplasmic incompatibility": males with Wolbachia can only reproduce successfully with females carrying the same bacterium. The World Mosquito Program, funded by the Gates Foundation, reported a 77% reduction in dengue cases in trial areas in Indonesia.
Genetic Control Methods Comparison
CRISPR Gene Drive
Permanent change, self-spreading, targets Anopheles (malaria). Still in laboratory stage.
Oxitec (Self-limiting)
Offspring die, doesn't spread, targets Aedes. Tested in Brazil and Florida.
Wolbachia
Bacterial infection, spreads naturally, reduces virus transmission. Active in 14+ countries.
⚖️ Ethical Questions and Ecological Risks
Combating diseases through genetic modification of insects isn't purely a technical matter. A gene drive, once released, cannot be recalled. If it spreads beyond its target or causes cascading ecosystem reactions, the consequences would be irreversible.
What happens if an entire mosquito species disappears? Mosquitoes serve as food for bats, birds, fish, and spiders. Males feed on nectar and pollinate plants. How much would the food chain be affected? Some ecologists argue that Anopheles gambiae could be replaced by other species without serious consequences. But no one is certain.
Dr. Drew Hammond emphasized that gene drives must function as tools alongside existing ones — bed nets, insecticides, vaccines — not as replacements. His Imperial team is conducting risk assessments in Tanzania and Burkina Faso, ensuring local community consent before any potential release.
🤔 The Central Dilemma
If a gene drive could save hundreds of thousands of lives annually, is it ethically acceptable to not use it? And if something goes wrong in an entire ecosystem, who bears responsibility?
🔮 Where This Technology Leads
Imperial's Transmission:Zero team estimates they could begin first field trials within the next 2-3 years, after completing safety testing. In Tanzania, a laboratory has already been established for creating and handling modified mosquitoes. There, parasites collected from locally infected students are used to confirm the modification works against real — not just laboratory — strains of Plasmodium.
Collaborators at the Institute for Disease Modeling of the Bill & Melinda Gates Foundation developed a predictive model showing gene drive technology could dramatically reduce malaria cases even in areas with high transmission intensity. Meanwhile, newer approaches — like precision-guided sterile insect technique (pgSIT) with CRISPR — create sterile male mosquitoes without gene drives, reducing the risk of irreversible environmental changes.
Malaria kills a child every two minutes. If genetic technology can change that, the question isn't whether we'll use it — but how.