Mice in an Emory University lab learn to fear the scent of cherry blossoms. Every time they smell it, they receive a mild electric shock. Then they breed — producing offspring that have never smelled this fragrance, never received a shock. Yet they tremble at the scent. Their grandchildren do too. Something passed from parent to child — but it wasn't a change in DNA. It was something above the DNA. It was epigenetics.
📖 Read more: What You Eat Changes Your Genes: Epigenetics Explained
What Is Epigenetics: The Code Above the Code
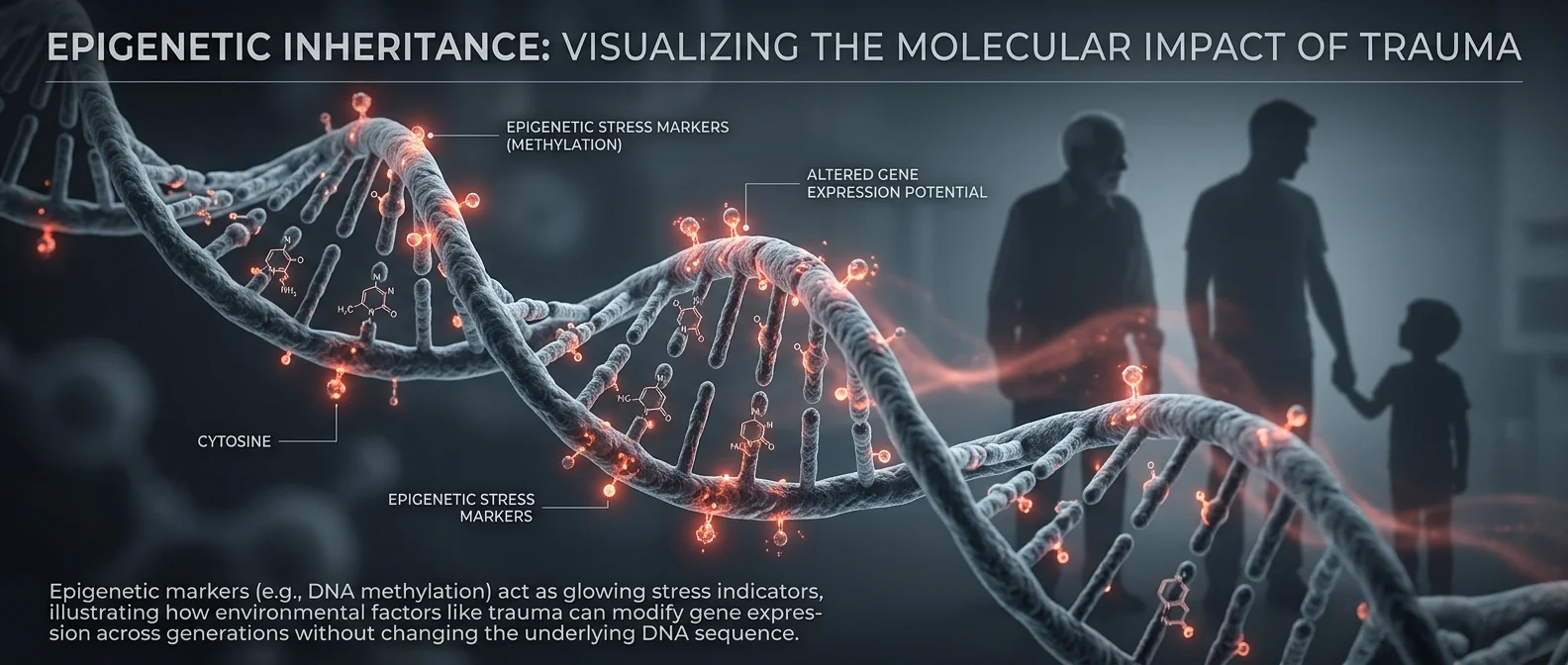
Your DNA doesn't change — but how it gets “read” changes dramatically. Epigenetics (epi = above) studies chemical modifications that turn genes on or off without altering the A-T-C-G base sequence. The three main mechanisms are: DNA methylation (adding -CH₃ groups to cytosine, mainly at CpG islands, which silences genes), histone modifications (acetylation “unfolds” chromatin and opens genes, methylation closes them), and non-coding RNAs (microRNAs that destroy mRNA before they translate into proteins).
Every cell in your body has exactly the same DNA — roughly 20,000 genes across 3.2 billion base pairs — yet a neuron is completely different from a liver cell. The difference? Epigenetics. Different genes activate in each cell type — about 4,000-8,000 genes are active in any given cell, the rest stay silent. What modern science discovered is explosive: these modifications aren't just determined during embryogenesis — they change throughout life, responding to diet, stress, toxins, even social experiences.
The Dias-Ressler Experiment: Inherited Fear
In 2014, Brian Dias and Kerry Ressler published a study in Nature Neuroscience that shocked biology. They exposed mice to acetophenone (cherry scent) combined with electric shock, until the animals developed fear of the smell. Then they bred them with mice that were never exposed. The children (F1) — with no previous experience — showed heightened startle responses to acetophenone, but not to other odors. Even more striking: the grandchildren (F2) reacted as well.
The biological explanation was equally stunning. The Olfr151 gene (encoding the M71 olfactory receptor for acetophenone) showed reduced methylation in the sperm of traumatized fathers — meaning it was more “open,” more active. The offspring had larger M71 glomeruli in their olfactory bulbs — more neurons dedicated to this exact scent. Fear information passed through chemical marks in sperm, without any change to the DNA sequence. The experiment extended to in vitro fertilization (IVF) — offspring born through IVF showed the same phenomenon, ruling out explanations through maternal behavior or shared environment.

Holocaust and Epigenetics: The Yehuda Study
Rachel Yehuda, professor of psychiatry at Icahn School of Medicine at Mount Sinai, has studied Holocaust survivors for decades. In 2016 she published a landmark finding in Biological Psychiatry: concentration camp survivors showed altered methylation in the FKBP5 gene — a crucial regulator of the HPA axis (hypothalamus-pituitary-adrenal), which controls cortisol response to stress.
📖 Read more: Why We Age: The Biology Behind Human Aging in 2026
Their adult children — born after the war, in safe environments, with no personal experience of persecution — showed the opposite methylation pattern in the same gene: hypermethylation instead of hypomethylation. The result was lower baseline cortisol levels, greater sensitivity to glucocorticoids, and triple the risk of PTSD. The study included 32 Holocaust survivors, 22 offspring, and control groups. The second generation never lived through the horror — but their bodies were “tuned” as if living under chronic threat.
The Dutch Hunger Winter: Famine That Passed Through Generations
During winter 1944-1945, Nazi occupation caused massive famine in western Netherlands — the “Hongerwinter” left 22,000 dead. Women who were pregnant during the famine gave birth to babies who — decades later — showed increased rates of obesity, type 2 diabetes, and cardiovascular disease. The Lumey group published that babies exposed to famine during the first three months of pregnancy had reduced methylation in the IGF2 gene (Insulin-like Growth Factor 2) — 60 years later.
Even more striking: the grandchildren of these women (third generation) also showed increased body weight and metabolic dysfunction — despite never experiencing hunger. The explanation? Famine “programmed” metabolism for maximum energy storage — a survival advantage under starvation conditions, but catastrophic in modern conditions of food abundance. And this program was inherited without any change to the DNA sequence. Similar findings emerged from studying the Swedish village of Överkalix — where food availability in grandfathers predicted cardiovascular mortality in grandchildren two generations later.

Maternal Care: Epigenetics in Action
Michael Meaney at McGill University (Montreal) showed that maternal care directly changes epigenetics. Rat mothers who lick and groom their newborns more create offspring with lower cortisol levels, less anxiety, and better memory — due to increased histone acetylation at the glucocorticoid receptor (NR3C1) in the hippocampus. Mothers who neglected their pups produced anxious adults with permanently high cortisol.
In human studies, autopsies of suicide victims who experienced childhood abuse revealed NR3C1 hypermethylation in the hippocampus — the exact same pattern as in rats. Suicide victims without abuse history had normal methylation — confirming the difference was epigenetic, not genetic. Love, literally, leaves a chemical imprint on the genome.
📖 Read more: Salamanders Regenerate Organs: Could Humans Do the Same?
Plants and Insects: Epigenetics Isn't Just Human
Transgenerational epigenetic inheritance is particularly strong in plants. Arabidopsis thaliana (mustard plant) carries epigenetic changes for over 30 generations — much more stable than mammals, since plants don't “reprogram” their epigenetic marks as drastically during reproduction. Aphids switch between winged and wingless forms through epigenetic changes — responding to population pressure without genetic mutation. In C. elegans worms, epigenetic changes caused by high temperature have been recorded for 14 generations, through small RNA mechanisms transmitted via sperm and eggs.
Criticisms and Limits: What We Still Don't Know
Epigenetic inheritance remains controversial in humans. The Yehuda and Dutch Hunger Winter studies can't completely eliminate "confounding factors": socioeconomic conditions, maternal behavior, shared environment. In mammals, epigenetic marks get “erased” twice: once during gamete formation (gametogenesis) and once immediately after fertilization. This “reprogramming” should theoretically delete trauma marks — but some imprinted genes (like IGF2, MEST, PEG3) escape.
Interpretations presenting epigenetics as “neo-Lamarckism” (inheritance of acquired characteristics) are oversimplified and scientifically inaccurate. Lamarck believed using an organ strengthens it and passes it on — epigenetics works completely differently. It doesn't invalidate Darwinian evolution through natural selection — it complements it, offering an additional regulatory layer that allows faster (but usually temporary) adaptation to environmental changes, especially during critical developmental windows.
Clinical Applications and the Future
Epigenetic therapy is already developing and entering clinical practice. DNA methyltransferase inhibitors (azacitidine, decitabine) are already used in myelodysplastic syndromes and acute myeloid leukemia. HDAC inhibitors (vorinostat, romidepsin) were FDA-approved for T-cell lymphomas. The idea is attractive: if epigenetic changes are reversible (unlike genetic mutations), then trauma could theoretically be “healed” at the molecular level — not just through psychotherapy, but with targeted pharmacology that “reopens” or “closes” specific genes.
Ethical questions follow: if poverty, violence, pollution leave permanent epigenetic marks on offspring, then social injustice acquires a biological dimension — and responsibility shifts from “individual” to “structural.” Epigenetics taught us something deeply human: one generation's experiences get written into the biology of the next.
Sources:
- Dias, B.G. & Ressler, K.J. (2014). "Parental olfactory experience influences behavior and neural structure in subsequent generations." Nature Neuroscience, 17(1), 89-96. DOI: 10.1038/nn.3594
- Yehuda, R., Daskalakis, N.P., et al. (2016). “Holocaust Exposure Induced Intergenerational Effects on FKBP5 Methylation.” Biological Psychiatry, 80(5), 372-380. DOI: 10.1016/j.biopsych.2015.08.005