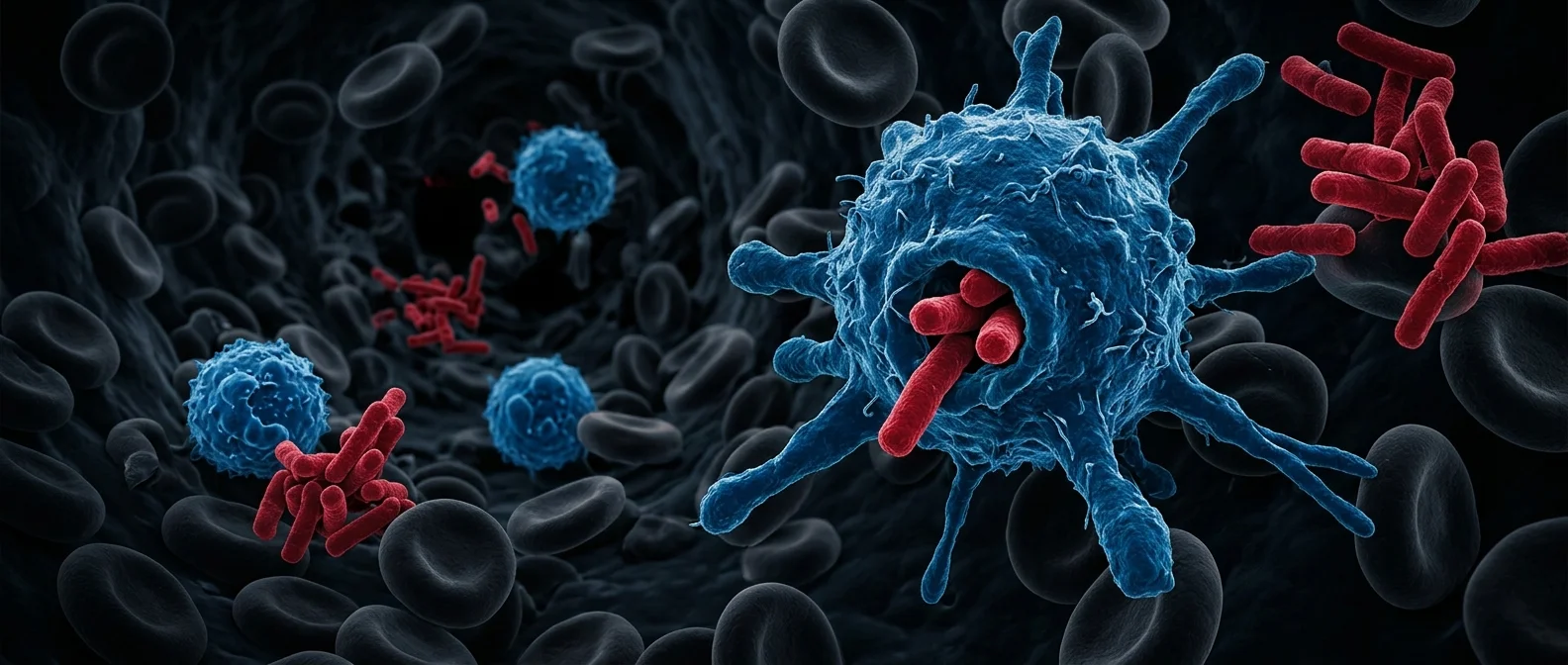
Right now, as you read these lines, a war is raging inside your body. You can't hear it, you can't feel it, but millions of cells are scanning every drop of blood, every tissue, every entry point. Bacteria, viruses, fungi — invaders constantly breaching the barriers of skin and mucous membranes. White blood cells are the army that stops them. And the way they work resembles an organized military operation more than a biological function.
📖 Read more: How Babies Acquire Billions of Bacteria in 72 Hours
The Invisible Army: Numbers and Ranks
In the blood of a healthy adult, between 4,000 and 11,000 white blood cells circulate per microliter at any given moment. Multiply this number by the approximately 5 liters of blood in your body, and we're talking about tens of billions of cells in permanent patrol — along with a massive reserve stockpile in bone marrow, spleen, and lymph nodes. Each type of white blood cell has a specific role — like different corps of an army. Neutrophils, macrophages, lymphocytes, eosinophils, basophils, and dendritic cells: each category specializes in different threats and communicates with the others.
Russian biologist Ilya Metchnikoff was the first to observe these cells in action in 1882, when he stuck a thorn into a starfish larva and watched cells gather around the invader. He named them phagocytes — “cells that eat” — and his discovery is considered the birth of immunology. He won the Nobel Prize in Medicine in 1908, along with Paul Ehrlich who studied antibodies.
Neutrophils: The First Line
If white blood cells are an army, neutrophils are the infantry. They make up 60-70% of all white blood cells and are the first to arrive at the infection site — usually within minutes. How do they know where to go? Damaged cells and bacteria release chemical signals — cytokines and chemokines — that function like “flares” on the battlefield. Neutrophils follow the chemical gradient, moving toward the highest signal concentration through a process called chemotaxis.
Once they arrive, neutrophils use three methods of destruction. First, phagocytosis: they engulf the invader and dissolve it with toxic enzymes inside specialized vesicles. Second, degranulation: they release antimicrobial substances into the surrounding space. Third — and most dramatic — NETs (Neutrophil Extracellular Traps): neutrophils literally explode, casting out their DNA like a net that traps bacteria. They commit suicide to save the organism.

📖 Read more: Microbiome: 38 Trillion Bacteria That Control Your Body
Macrophages: The Sweepers That Never Forget
If neutrophils are the kamikazes, macrophages are the experienced generals. They live much longer — weeks or even months — and aren't confined to blood. They're found in every tissue: in lungs (alveolar macrophages), liver (Kupffer cells), brain (microglia). Every tissue in the body has its own permanent guards.
Macrophages don't just destroy — they communicate. After phagocytosing an invader, they break down its proteins into small pieces (peptides) and “present” them on their surface through MHC class II molecules. They function like spies bringing back photographs of the enemy: showing T-lymphocytes exactly who the target is. Without this antigen presentation step, specific adaptive immunity cannot activate — the army doesn't know who it's hunting. This is the bridge between innate and acquired immunity, two systems working in perfect harmony.
T-Lymphocytes: The Targeted Assassins
T-cells are born in bone marrow but mature in the thymus gland — hence the “T.” There, they undergo strict "training": the thymus tests whether each T-cell recognizes foreign antigens but NOT your own cells. Those that fail — about 95% — are destroyed on the spot.
CD8+ cytotoxic T-cells are the executioners. They recognize infected cells through MHC class I molecules and kill them by secreting perforin, a protein that opens pores in their membrane, and granzymes, serine enzymes that trigger apoptosis — programmed cell death. Once destruction is complete, the T-cell withdraws and searches for the next target. It can kill multiple infected cells sequentially without being destroyed itself.
CD4+ helper T-cells don't kill directly — instead, they coordinate the entire immune response, activating B-cells, macrophages, and cytotoxic T-cells through cytokines. HIV destroys precisely these cells, explaining why the disease dismantles the entire immune system: without the “generals,” the rest of the army loses coordination.
📖 Read more: Sea Turtles Use Earth's Magnetic GPS to Navigate 1000s Miles
B-Lymphocytes: The Antibody Factories
If T-cells kill, B-cells arm. Each B-lymphocyte carries antibodies on its surface specific to only one antigen. When a B-cell encounters the invader it recognizes, it activates and transforms into a plasma cell — a small factory that secretes thousands of antibodies per second.
Antibodies don't kill on their own in most cases. They stick to invaders and “mark” them — a process called opsonization. Marked bacteria become much easier targets for phagocytes, which recognize them through specialized Fc receptors. Meanwhile, antibodies can directly neutralize toxins and prevent viruses from entering cells by physically blocking entry receptors on the cell membrane. A single plasma cell can secrete about 2,000 antibodies per second — a production rate few biological factories can match.

Inflammation: The War You Can See
Swelling, redness, heat, pain — the four classic signs of inflammation described by Roman physician Celsus 2,000 years ago. These aren't symptoms of disease. They're proof the army is working. Blood vessels dilate (redness, heat), their permeability increases (swelling), and nerve endings activate (pain) — all to get more white blood cells to the site faster.
Histamine, released by mast cells and basophils, is a key trigger of this reaction. Antihistamine drugs don't kill microbes — they simply calm some mechanisms of war. Which is sometimes necessary, because if inflammation spirals out of control — as in sepsis or cytokine storm — it can become as deadly as the invader itself. The body's ability to regulate inflammation intensity is one of the most critical balances in biology.
📖 Read more: Sign Language Chimps: Whatever Happened to Them?
Immunological Memory: How the Army Remembers
After each battle, a small percentage of T and B-cells don't die. They transform into memory cells — long-lived specialist guards that circulate quietly in blood and lymph nodes, patiently waiting for the same invader to appear again. Some memory cells survive decades — even an entire lifetime. If the same pathogen appears, the secondary immune response is explosively faster and stronger: instead of weeks, it takes only hours. This explains why you catch a cold once from a specific flu strain but rarely twice.
This mechanism is the basis of every vaccine. A vaccine doesn't give medicine — it gives information. It presents the immune system with a harmless piece of the enemy (protein, weakened virus, mRNA) so memory cells are created before infection. A war rehearsal without danger.
Autoimmune: When the Army Turns Against Itself
What happens when the guard loses its bearings? Autoimmune diseases — lupus, rheumatoid arthritis, multiple sclerosis, type 1 diabetes — are exactly that: the immune system attacks your own tissues as if they were hostile. In lupus, antibodies turn against nuclear DNA. In type 1 diabetes, T-cells destroy the pancreatic β-cells that produce insulin.
The exact reason the immune system gets “confused” remains a subject of intensive biomedical research. Genetic predisposition through HLA genes, environmental factors like infections and stress, molecular mimicry (when a bacterium carries proteins chemically similar to host tissues) — it's likely a combination of multiple causes. The army that keeps you alive can, under certain unfortunate circumstances, become your most dangerous enemy. The fine line between immunological protection and self-destruction remains one of the greatest challenges in modern immunology.
Sources:
- Metchnikoff, “Immunity in Infective Diseases,” Cambridge University Press, 1905
- Janeway & Medzhitov, “Innate Immune Recognition,” Annual Review of Immunology, 2002