Imagine a tiny pair of “scissors” that can cut DNA at exactly the right spot, remove a defective gene, and replace it with a healthy one. This is no longer science fiction — it's CRISPR-Cas9 technology, already curing diseases once thought to be incurable.
📖 Read more: Xenotransplantation: Animal Organs in Humans
What Is CRISPR?
CRISPR (Clustered Regularly Interspaced Short Palindromic Repeats) is a genetic engineering technique that allows modification of living organisms' genomes with pinpoint accuracy. It's based on a natural defense system found in bacteria: the CRISPR-Cas9 system, which bacteria use to destroy viruses that infect them.
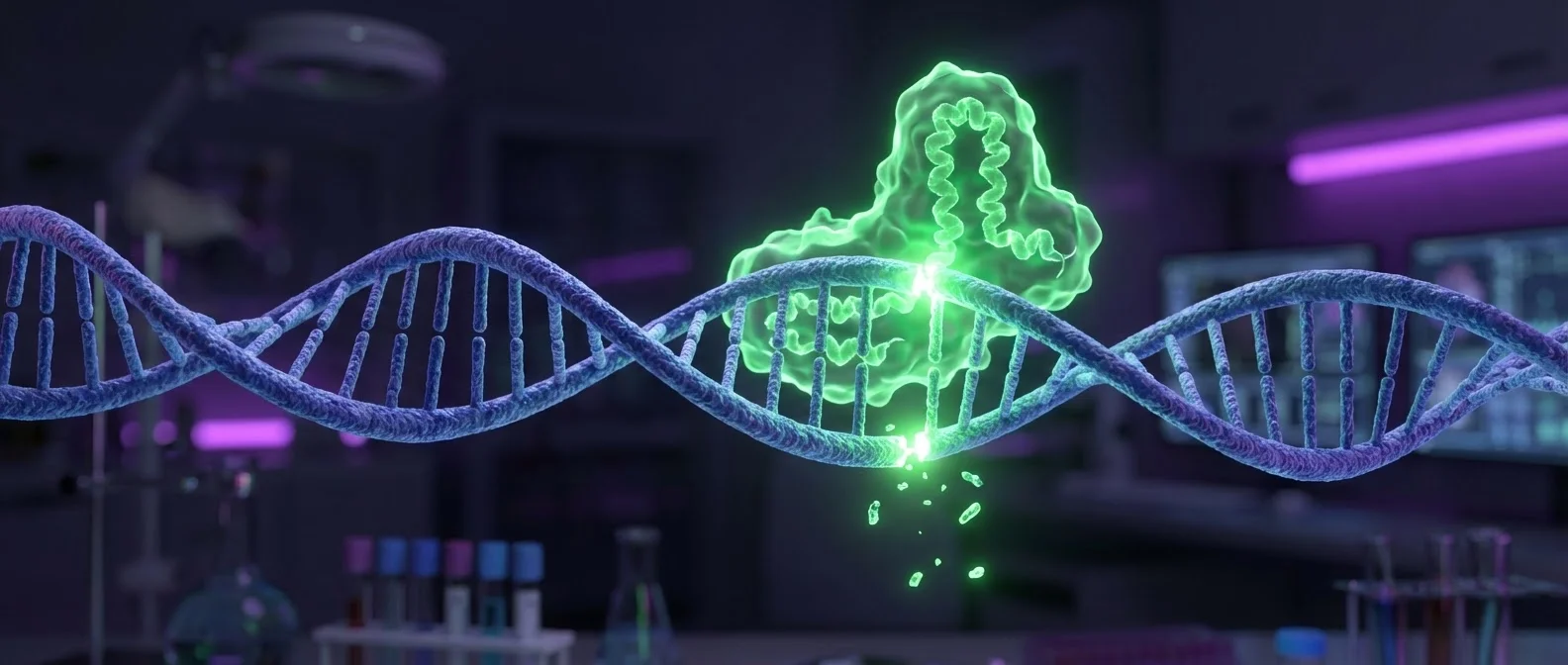
The Cas9 protein functions as molecular “scissors.” Combined with a synthetic guide RNA (gRNA), it can locate a specific DNA sequence, cut it, and then the cell's natural repair mechanisms take over to “fix” or replace the damage. Unlike older methods (ZFNs, TALENs), it works quickly and costs a fraction of the price.
🧬 How Does It Work?
Guide RNA locates target → Cas9 cuts the DNA double helix → Cellular mechanisms repair or replace → Gene corrected. The precision is so great it can target a single gene among 3 billion base pairs.
The History of a Revolution
"There is enormous power in genetic engineering. The question isn't whether we can, but whether we should."
Casgevy: The First CRISPR Drug
In November 2023, the UK's regulatory authority MHRA approved Casgevy (exagamglogene autotemcel) — the world's first drug based on CRISPR-Cas9 technology. Developed by Vertex Pharmaceuticals and CRISPR Therapeutics for treating two serious blood diseases: sickle cell disease and beta-thalassemia.
🩸 How It Works
Modifies the patient's hematopoietic stem cells to produce fetal hemoglobin (HbF), bypassing the defective gene.
✅ Effectiveness
29 of 31 patients (93.5%) required no transfusions for 12+ months. None experienced graft rejection.
🌍 Approvals
United Kingdom (November 2023), Bahrain (December 2023), FDA (December 2023), European Union (February 2024).
💰 Cost
$2.2 million in the US, ~£1 million in the UK. Single dose, lifetime result.
Diseases CRISPR Is Changing
There are approximately 6,000 known genetic diseases, most of them incurable. CRISPR is opening therapeutic pathways for many:
Prime Editing: The Next Generation
While classic CRISPR-Cas9 cuts the DNA double helix (risking errors), Prime Editing introduces modifications without cutting both strands. David Liu (MIT) developed this method that can theoretically correct 89% of known genetic defects.
Meanwhile, the PASTE technique (2022) enables insertion of massive DNA segments — up to 36,000 base pairs — without problematic double-strand breaks, opening pathways for treating multi-mutation diseases.
📊 CRISPR as a Diagnostic Tool
Beyond treatment, CRISPR is also used in diagnostics. The SHERLOCK and AIOD-CRISPR systems detect viruses (including SARS-CoV-2) with accuracy down to 10 copies per microliter — in portable, affordable devices.
CRISPR in Agriculture
CRISPR has jumped from hospitals to farms. In September 2021, the first CRISPR foods went on sale in Japan — tomatoes with 5x more GABA, a calming amino acid. Fish growing to twice their natural size followed.
The technology is being applied to rice, wheat, soybeans, and cacao — for increased yield, disease resistance, reduced allergens (gluten-free wheat), improved nutritional value, and extended shelf life.
Ethical Dilemmas & Risks
⚠️ Off-Target Effects
CRISPR can “cut” wrong spots in DNA — off-target changes represent the greatest risk. Newer versions (Cas9 nickases, base editors) significantly reduce this risk.
👶 Germline Modification
Modifying human embryos (germline editing) is inherited generation to generation. In 2018, He Jiankui created the first CRISPR babies — he was sentenced to 3 years in prison.
💊 Access & Cost
At $2.2 million per treatment, who will have access? Health inequality could deepen, creating “genetic haves and have-nots.”
🧪 Bioethics & Eugenics
If we can “fix” diseases, can we “enhance” traits? The line between therapy and eugenics grows increasingly blurred.
Global Impact
Beta-thalassemia is particularly prevalent in Mediterranean populations, parts of Africa, and Southeast Asia. Approximately 1.5% of the global population are carriers. The approval of Casgevy by the EU (February 2024) and FDA opens a new era for thousands of patients worldwide who depend on regular blood transfusions.
Research institutions across the globe — from the Broad Institute and MIT to universities in Europe and Asia — are racing to expand CRISPR applications. Over 80 clinical trials were underway by 2024, targeting everything from blood disorders to solid tumors.
"The New Biology of CRISPR could be the greatest tool invention since primitive humans created the first tools."
What Lies Ahead?
CRISPR is moving faster than anyone expected. New versions (Cas12, Cas13, base editing, prime editing) are expanding capabilities into RNA editing, epigenomics, agricultural technology, and biosecurity. Clinical trials are multiplying — over 80 were underway in 2024.
By 2030, analysts predict CRISPR therapies for muscular dystrophy, cystic fibrosis, Huntington's disease, and certain cancers. By 2040, gene therapy could become as routine as antibiotics are today.
The real challenge is no longer technological — it's social, ethical, and economic. If we can eliminate hereditary diseases, who does that gift belong to? The answer will determine whether CRISPR becomes the greatest medical revolution — or the greatest driver of inequality.