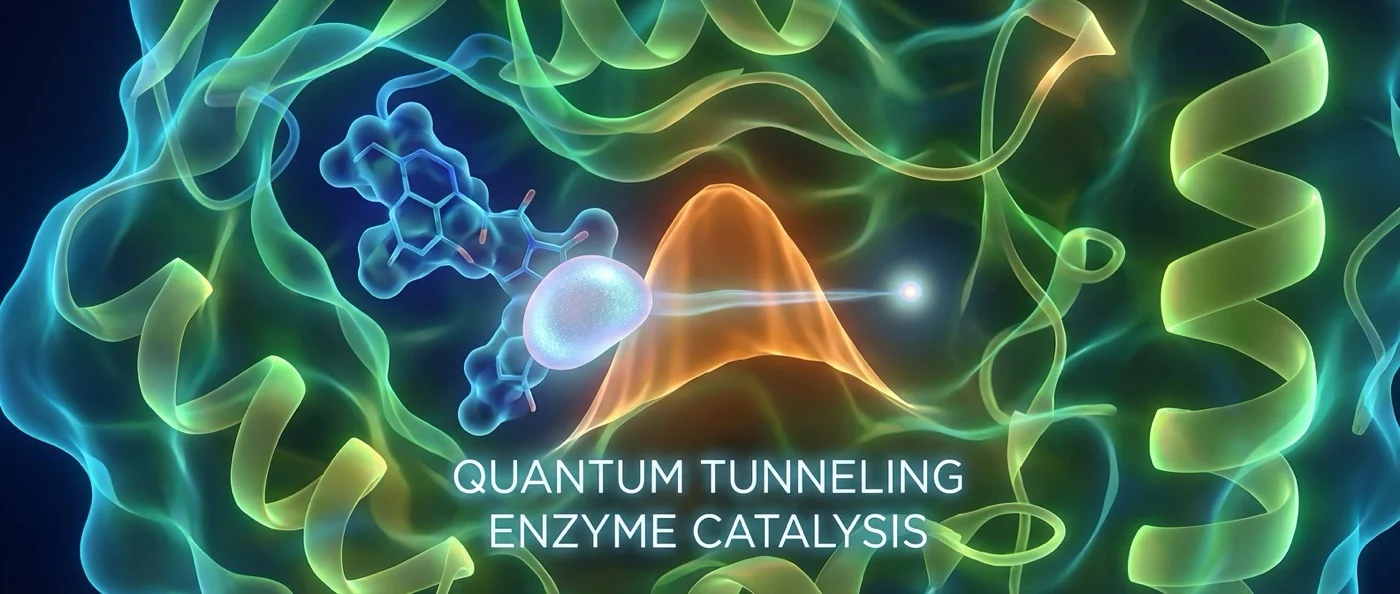
Studies show that enzymes use quantum tunneling to transfer hydrogen atoms. Without quantum phenomena, biological catalysis would be far too slow for life.
⚗️ Enzymes — the invisible accelerators of life
Every second, billions of chemical reactions take place inside our cells. Digestion, respiration, DNA synthesis — none of this would be possible without enzymes, the proteins that accelerate biological reactions by millions of times. In classical biochemistry, enzymes work by lowering the so-called “activation barrier” — the minimum energy a reaction needs to get started. But there is a mystery: some enzymes operate faster than classical theory predicts. The explanation lies in quantum mechanics.
🔀 Quantum tunneling — a shortcut through the impossible
In the classical world, if a ball doesn't have enough energy to pass over a wall, it stops. In the quantum world, things are different. Particles — protons, electrons, hydrogen ions — simultaneously behave as waves. This wave-like nature allows them to “pass through” energy barriers that classically would be impenetrable. The phenomenon is called quantum tunneling.
In enzymes, this happens at distances on the order of 1 Ångström (10⁻¹⁰ meters) — roughly the length of a chemical bond. Hydrogen ions (H⁺) and electrons don't need to “climb over” the energy barrier. They can penetrate it quantum mechanically — as if passing through the wall instead of jumping over it.
🔬 The discovery — first evidence
The story began in 1966, in a laboratory studying photosynthetic bacteria. Don DeVault and Britton Chase studied the bacterium Chromatium and observed something remarkable: at temperatures below 100 K (−173 °C), the rate of cytochrome oxidation did not decrease — it was temperature-independent. Classically, lower temperature means less kinetic energy and slower reactions. The explanation? Electrons weren't passing “over” the barrier — they were going “through” it via quantum tunneling.
Three decades later, Judith Klinman at UC Berkeley provided the critical proof. In 1994, a study on soybean lipoxygenase revealed “extremely large kinetic isotope effects” (KIE). When ordinary hydrogen is replaced by deuterium — a heavier isotope — the reaction slows down. Classically, the slowdown should be modest (KIE ≈ 7). In the lipoxygenase, however, the slowdown was 80 times greater — far beyond any classical prediction. This was the clear fingerprint of quantum tunneling.
⚛️ Masgrau 2006 — the atomic description
The fullest confirmation came in April 2006, in a publication in the leading journal Science. Laura Masgrau's team, together with Roujeinikova, Johannissen and colleagues, achieved something unprecedented: they described at the atomic level an enzymatic reaction entirely dominated by proton tunneling.
The enzyme was aromatic amine dehydrogenase (AADH) during the oxidation of tryptamine. Combining X-ray crystallography, kinetic measurements and computational simulations, the team showed that proton transfer does not follow classical transition state theory. Tunneling was not a minor correction — it was the dominant mechanism of the reaction.
Why it matters: The Masgrau et al. (2006) study in Science showed that quantum proton tunneling can be the dominant reaction mechanism in certain enzymes — not a small quantum correction, but the very basis of the entire catalysis.
This principle holds broadly. The review by Nagel and Klinman (2006) in Chemical Reviews concluded that quantum tunneling plays a significant role in hydride transfer across many enzymes in living organisms.
⚡ Mitochondria — the quantum energy machine
If enzymes are the cell's workers, mitochondria are its power stations. The electron transport chain (ETC) converts food biomass into ATP — the energy currency of life. And in this process too, quantum tunneling plays a starring role.
The inner mitochondrial membrane is just ~7.5 nm thick. Within it, electrons penetrate via quantum tunneling through Complex I (NADH:ubiquinone oxidoreductase) and Complex III (CoQH₂-cytochrome c reductase). The thermodynamic efficiency of this conversion reaches 60-70% — far superior to any human-made engine.
Ferritin — an iron-storage protein found in plants and animals — exhibits quantum electron tunneling over distances up to 8 nm at room temperature. This tunneling is temperature-independent — exactly as DeVault first hypothesized in 1966 — suggesting it is essentially coherent. A remarkable quantum phenomenon in a warm, noisy biological environment.
🧬 Broader implications — quantum biology
Quantum tunneling in enzymes is not an academic curiosity — it is likely critical to the very existence of life. Without tunneling, the rate of many enzymatic reactions would be ~1000 times slower. Slow enough that biological catalysis could not sustain metabolism.
"Quantum biology poses a fascinating question: does life exploit quantum phenomena in non-trivial ways?" — Jim Al-Khalili, physicist, TED Talk 2015
Beyond enzymes, quantum tunneling is implicated in DNA mutations. Proton transfer between bases can cause tautomeric shifts — changes in nucleotide structure that lead to misreplication. A study by Slocombe, Al-Khalili and Sacchi (2021) in Physical Chemistry Chemical Physics showed that quantum proton tunneling can cause point mutations in AT and GC base pairs. Quantum mechanics doesn't just power life — it may drive the very mutations on which natural selection acts.
Today the field of quantum biology is evolving rapidly. Researchers are studying how understanding tunneling could help in the design of new drugs — if we know exactly how an enzyme uses quantum tunneling, we can design inhibitors that target this mechanism. Quantum computational chemistry opens new avenues in this direction.
From the photosynthetic bacteria that DeVault studied to the mitochondria in every one of our cells, quantum tunneling lies at the heart of biological catalysis. Our enzymes don't operate “despite” physics — they operate within it, exploiting the most paradoxical properties of the quantum world.