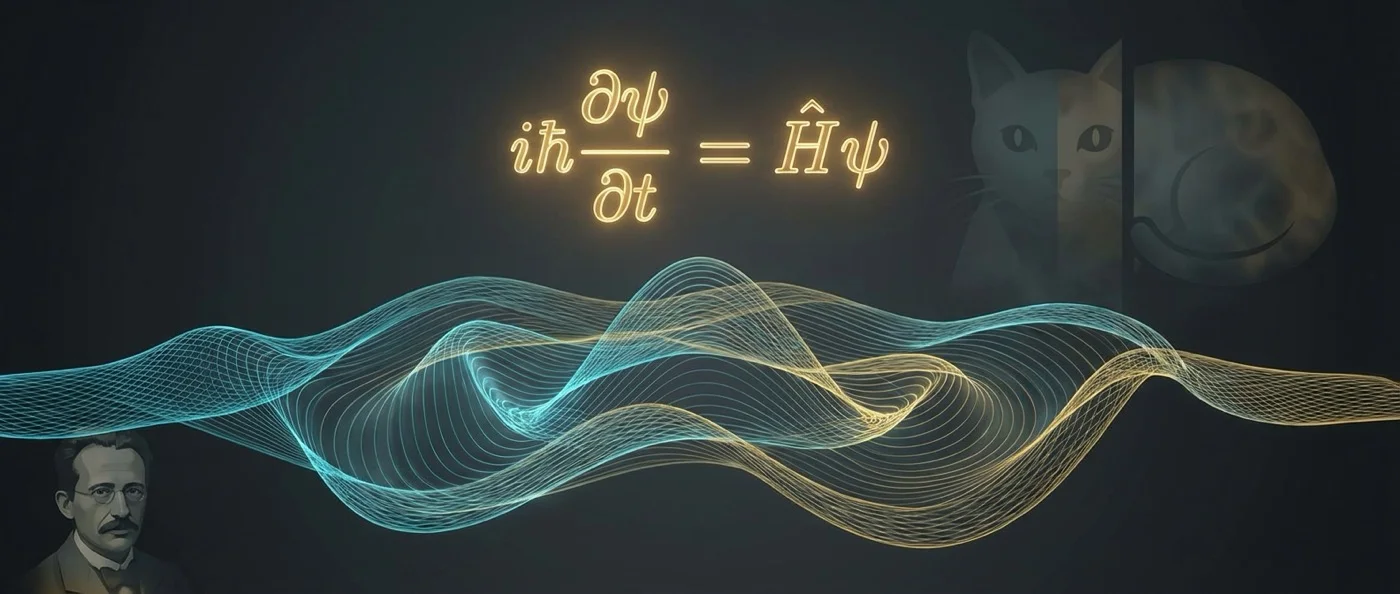The Schrödinger equation describes how the wave function evolves. Who was this Austrian physicist who hated the Copenhagen interpretation — yet gave birth to its foundation?
🏔️ A sanatorium in the Alps, December 1925
Erwin Schrödinger did not discover the most important equation in quantum physics inside a university laboratory. He found it while recovering from tuberculosis at a sanatorium in Arosa, Switzerland, surrounded by snow-covered peaks — and most likely not alone. Biographer Walter Moore notes that Schrödinger had with him a “mysterious girlfriend” whose identity remains unknown. Within a few weeks, the equation that would change everything was born.
🎓 From Vienna to Zurich
Erwin Rudolf Josef Alexander Schrödinger was born on August 12, 1887 in Vienna. He was an only child — his father Rudolf was a botanist and his mother Georgine was half Austrian, half English. He studied physics at the University of Vienna under Franz Exner and Friedrich Hasenöhrl, and received his doctorate in 1910.
After military service in World War I, his academic career led him to the University of Zurich in 1921. There, in November 1925, he attended a lecture on Louis de Broglie's matter wave theory. Physicist Peter Debye made a comment that seemed innocent: “If particles behave as waves, they must obey some sort of wave equation.” Schrödinger took that thought literally.
Schrödinger did not start from scratch. He was guided by the mathematical analogy between mechanics and optics that William Hamilton had discovered in the 19th century: particle mechanics is to wave optics what geometric optics is to wave optics — a limiting case. This insight showed him the way.
📜 Four publications in five months
In January 1926, Schrödinger published in the journal Annalen der Physik the first of four historic papers titled “Quantisierung als Eigenwertproblem” (Quantization as an Eigenvalue Problem). In it, he presented his equation and showed that it yields the correct energy levels for the hydrogen atom — in full agreement with experimental data.
Just four weeks later, he published a second paper solving the quantum harmonic oscillator and the rigid rotor. In May, a third followed, in which he proved that his wave mechanics is mathematically equivalent to Werner Heisenberg's matrix mechanics. Finally, a fourth paper introduced the time-dependent equation — the one we use everywhere today.
📐 What the equation says
The Schrödinger equation is to quantum mechanics what Newton's second law is to classical physics. Newton says: "Give me the position and velocity of an object now, and I'll tell you where it will be tomorrow." Schrödinger says something analogous: "Give me the wave function Ψ of a system now, and I'll tell you how it will evolve in the future."
The equation describes how the wave function Ψ(x,t) — a mathematical entity that assigns a complex number to each point in space and each moment in time — changes over time. It is linear, meaning that if two solutions are valid, then any linear combination of them is too — this is the mathematical basis of quantum superposition.
It is also "unitary": it preserves total probability equal to 1. Whatever begins as 100% probability, remains 100%. Information is never lost within the equation — a fact that later triggered enormous discussions about black holes and the information paradox.
😤 “I don't like it, and I'm sorry I ever had anything to do with it”
Schrödinger believed that the wave function Ψ represented something real — initially he thought |Ψ|² was an electric charge density. But Max Born, that same year, proposed a radically different interpretation: |Ψ|² is not charge, but probability. The wave function doesn't tell you where a particle “is” — it tells you where it might be found if you look.
Schrödinger reacted with fury. “I don't like it, and I'm sorry I ever had anything to do with it,” he declared later. When the Göttingen physicists — Heisenberg, Born, Jordan — began using his waves to calculate their matrices, Schrödinger reportedly shouted: "Now those damned Göttingen physicists are using my beautiful wave mechanics to calculate their shitty matrix elements!"
🐱 The cat, entanglement, the legacy
In 1933, Schrödinger shared the Nobel Prize in Physics with Paul Dirac “for the discovery of new productive forms of atomic theory.” He had already left Germany due to his opposition to Nazism.
In 1935, corresponding with Einstein, he constructed the famous thought experiment with the cat — dead and alive simultaneously — to ridicule the Copenhagen interpretation. Paradoxically, it became the most popular symbol of the very theory he hated. That same year, he published the paper that codified the concept of quantum entanglement — what he called "the characteristic that forces the entire departure from classical lines of thought."
In 1944, in Dublin, he wrote the book What Is Life?, in which he predicted that genetic information is stored in an “aperiodic crystal.” James Watson later stated that this very book inspired him in the discovery of the DNA structure in 1953.
Schrödinger died on January 4, 1961 in Vienna, at the age of 73, from tuberculosis — the same disease that had sent him to the sanatorium where his equation was written. At his grave in Alpbach, Austria, engraved on a circular plaque, lies the equation: iℏΨ̇ = HΨ
🔬 Why it changes everything
The Schrödinger equation is not merely a historical relic. Every atomic orbital in chemistry, every semiconductor in your phone, every laser in the supermarket, every qubit in a quantum computer — none of these would exist without it. From the moment Schrödinger published it, the wave function became the language with which nature is described at its most fundamental scale.
The deterministic evolution of the wave function, combined with its probabilistic “collapse” during measurement, constitutes the so-called “measurement problem” — perhaps the deepest open question in physics. Schrödinger himself proposed in 1952 that "the different terms of a superposition are not alternatives but all really happen simultaneously" — an idea that Hugh Everett later named the many-worlds interpretation.
An Austrian physicist who hated what he created. An equation written in a sanatorium. A cat that was never killed. And the entire modern world built upon them.