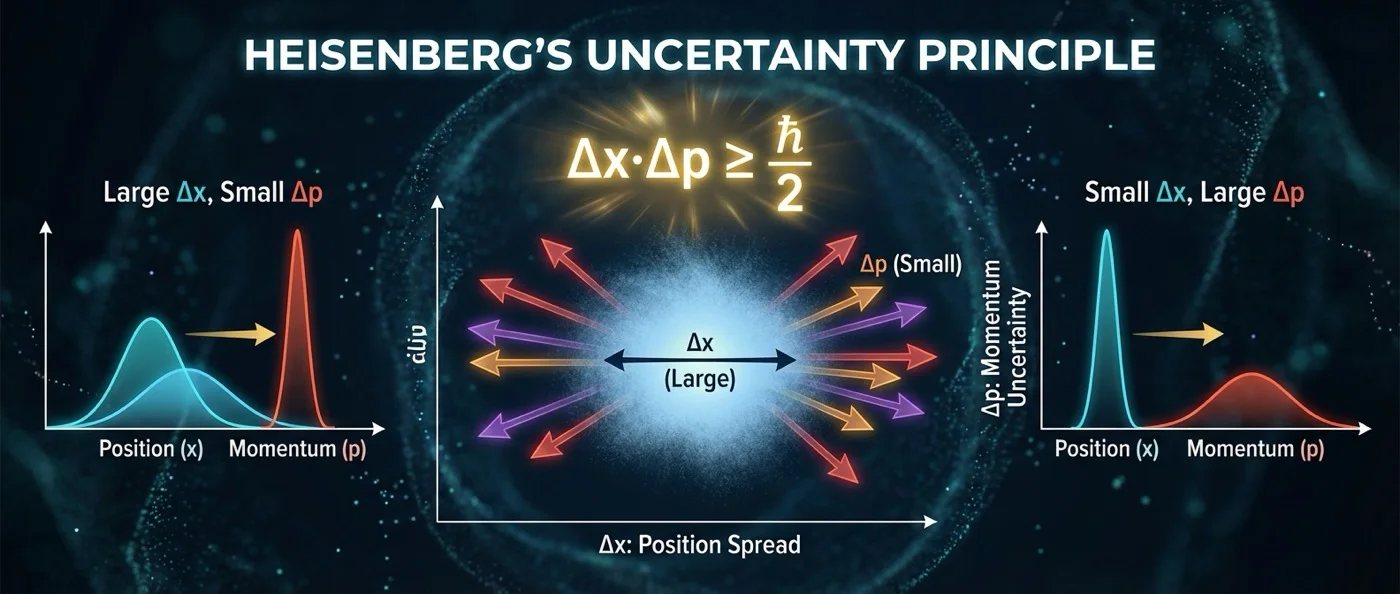
There is no way to simultaneously know the position and momentum of a particle. This is not a technological limitation — it is a fundamental law of nature.
🌐 A strange world with built-in limits
It sounds like a technical problem. As if we're saying: “our instruments aren't precise enough.” But Heisenberg's uncertainty principle says something far more radical. It says that nature itself does not simultaneously possess a precise position AND a precise momentum for a particle. It's not that we cannot measure — it's that there is nothing there to be simultaneously measured.
This is perhaps the most subversive message of quantum mechanics. And certainly the one the world struggled most to accept — even physicists of Einstein's caliber.
🏝️ The young physicist on Heligoland
In June 1925, 23-year-old Werner Heisenberg fled to the island of Heligoland in the North Sea, escaping the pollen that was worsening his hay fever. There, between rock climbing and memorizing poems by Goethe, he made a discovery that would change physics forever.
Late at night, at three in the morning, he completed calculations showing that quantum quantities do not commute — the order in which you measure position and momentum matters. He was so shaken that he couldn't sleep. He waited for the sunrise on top of a rock.
That paper, the famous Umdeutung (Reinterpretation), was published in September 1925 and together with Max Born and Pascual Jordan established matrix mechanics — the first rigorous mathematical formulation of quantum mechanics. The central idea was simple but revolutionary: position X and momentum P are not numbers but matrices, and the order of their multiplication matters. Mathematically: XP − PX = iℏ, where ℏ is the reduced Planck constant.
📐 The formula that changed philosophy
In February 1927, Heisenberg wrote to Wolfgang Pauli a letter where he described for the first time what we now call the uncertainty principle. In the historic paper “Über den anschaulichen Inhalt der quantentheoretischen Kinematik und Mechanik” (On the Perceptual Content of Quantum Theoretical Kinematics and Mechanics), he used the term “Ungenauigkeit” (imprecision). Only in the footnote did the word “Unsicherheit” (uncertainty) appear — and later he always used “Unbestimmtheit” (indeterminacy).
The rigorous mathematical inequality was proved the same year (1927) by Earle Hesse Kennard:
The standard deviation of position (σx) multiplied by the standard deviation of momentum (σp) can never fall below ℏ/2. The more precisely you know the position, the more uncertain the momentum becomes — and vice versa. There is no way to “cheat” this constraint.
🔍 Why the measurement is NOT to blame
A common misconception is that the uncertainty arises because measurement “disturbs” the particle. This is the so-called observer effect, but it is not the uncertainty principle.
Heisenberg himself initially used a thought experiment — a gamma-ray microscope — to explain the principle. If you shoot a short-wavelength photon at an electron, you can measure its position accurately. But the photon transfers enormous momentum and “pushes” the electron in an uncertain way. Conversely, a long-wavelength photon doesn't disturb the momentum much, but doesn't reveal the position precisely.
However, Niels Bohr showed that this explanation wasn't entirely accurate. Uncertainty is an intrinsic property of every wave-like system. A wavefunction narrowly localized in space necessarily corresponds to a broad distribution in momentum space through the Fourier transform — and vice versa. This has nothing to do with measuring instruments. It is a mathematical necessity.
⏱️ Energy-time uncertainty
The principle is not limited to the position-momentum pair. An analogous relation exists for energy-time, though its precise formulation is more subtle. In 1945, Leonid Mandelstam and Igor Tamm proved that the duration of an energy measurement is related to its precision: ΔE · Δt ≥ ℏ/2.
This explains why unstable particles, like the Delta (Δ) particle, which lives only 10−23 seconds, have a broad mass distribution: ±120 MeV/c² around 1,232 MeV/c². Their brief existence is reflected in greater energy uncertainty.
🎲 “God does not play dice” — the great objection
Einstein never accepted that uncertainty is fundamental. He believed that randomness reflects our ignorance, not the nature of reality. In the historic EPR paper (Einstein-Podolsky-Rosen, 1935), he argued that quantum mechanics is “incomplete” — that there must be hidden variables simultaneously determining both position and momentum.
The Copenhagen Interpretation, championed by Bohr and Heisenberg, responded: there is no fundamental reality beyond what we can measure. The quantum state does not describe some “real” object but serves as a tool for predicting experimental outcomes.
Heisenberg himself was explicit: we can never know what a particle “really is.” We can only know numbers in tables that describe something about them.
💡 What it means for us
The uncertainty principle is not only philosophically significant. Today, quantum cryptography relies precisely on it: any eavesdropping attempt necessarily alters the measurement, because uncertainty does not allow complete knowledge without disturbance. Quantum computers operate exactly within this framework of uncertainty — exploiting superposition rather than fearing it.
Heisenberg won the Nobel Prize in Physics in 1932 “for the creation of quantum mechanics.” Later, he felt that Born and Jordan had not been adequately recognized. In a letter to Born, he admitted a “bad conscience” for receiving the prize alone. Born eventually received his own Nobel in 1954 — for the statistical interpretation of the wavefunction.
🌌 Nature sets its own boundaries
The uncertainty principle reminds us of something profound: the universe does not owe us transparency. It was not built to be fully understood. There are limits to knowledge that are not the result of human inadequacy, but architectural features of reality itself.
Nearly 100 years after Heisenberg's letter to Pauli, the uncertainty principle remains intact. No experiment has ever violated it. Every attempt at simultaneously measuring position and momentum with precision fails — not due to experimental limitations but fundamentally. The universe, at its deepest level, maintains an inalienable opacity. And perhaps that is the most important lesson quantum physics has taught us.