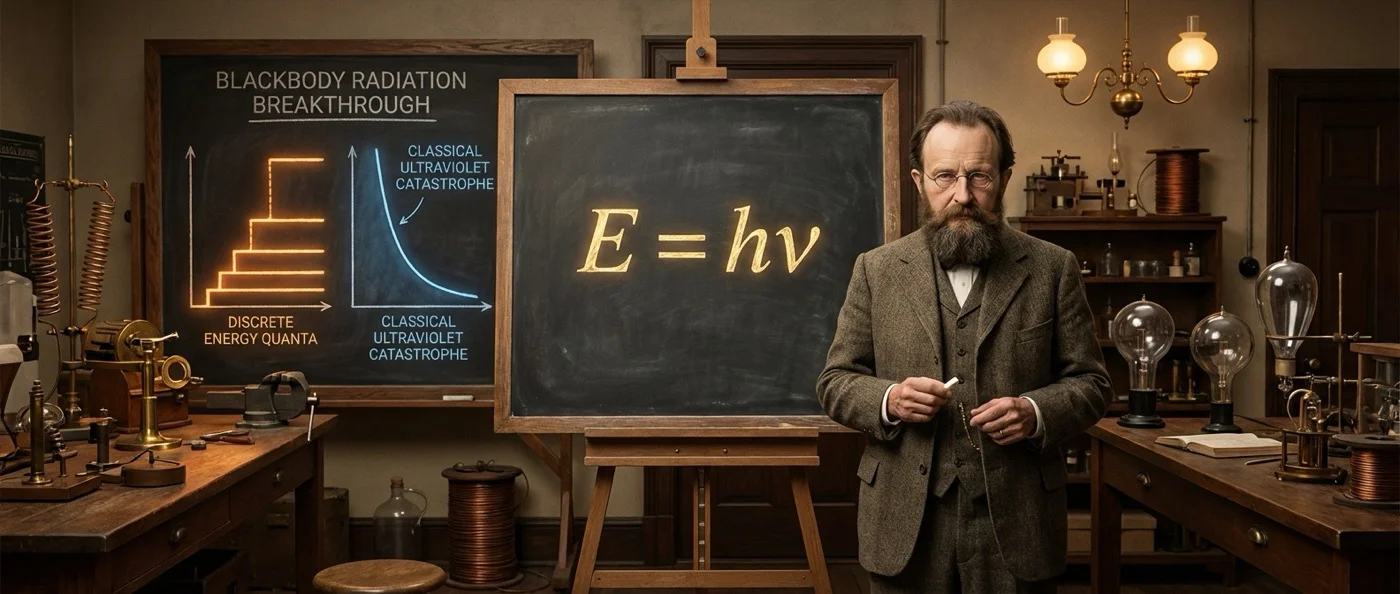At the end of the 19th century, a conservative German physicist did something he himself called “an act of desperation.” He introduced energy quanta as a mathematical trick — and, without meaning to, gave birth to an entirely new physics that overturned everything.
🌡️ What Was the Problem Troubling Physics at the End of the 1800s?
The problem was black-body radiation — how a hot object radiates energy. An ideal “black body” absorbs all radiation that falls on it and emits light according to its temperature. Gustav Kirchhoff formulated this challenge in 1859: how does the intensity of electromagnetic radiation depend on frequency and temperature?
The challenge was universal — the emission spectrum had to be described by a single law independent of the material. Kirchhoff himself was Planck's professor at the University of Berlin, and the search for this law — known as “Kirchhoff's challenge” — became the subject of decades of experimental and theoretical work. By the 1890s, experimentalists like Otto Lummer, Ferdinand Kurlbaum, and Heinrich Rubens had built precision cavity radiators and produced increasingly accurate measurements.
⚡ Why Did the Existing Laws Fail?
Two laws attempted to solve the problem — and neither fully succeeded.
Wien's law (1896) correctly predicted behavior at high frequencies (short wavelengths) but failed at low frequencies. Conversely, the Rayleigh–Jeans law gave correct results at long wavelengths, but at high frequencies it predicted that radiated energy tends to infinity — an absurdity that Paul Ehrenfest named the “ultraviolet catastrophe” in 1911.
The ultraviolet catastrophe was not a minor discrepancy. It predicted that every warm body would emit infinite energy — something obviously impossible. Classical physics, based on the equipartition theorem of energy, could not explain why high frequencies do not dominate the spectrum. The total energy integral simply diverged.
💡 How Did Planck Find the Solution — and Why Did He Call It 'an Act of Desperation'?
Max Planck had been working on the problem since 1894. He initially supported Wien's law, but on October 7, 1900, the experimentalist Heinrich Rubens informed him that new measurements at long wavelengths disagreed with it.
Within just a few days, Planck “patched together” the two formulas. On October 19, 1900, he presented an empirical formula to the German Physical Society (DPG) that fitted the data perfectly at every wavelength. Rubens immediately confirmed its agreement with experiment.
The problem was the physical interpretation. On December 14, 1900 — the date considered the “birth” of quantum theory — he presented a theoretical derivation. He used Boltzmann's statistical method, which he had previously rejected, and assumed that energy is quantized into discrete packets:
E = hν
Here ν is the frequency of radiation and h is the new constant — the Planck constant (h = 6.626 × 10⁻³⁴ J·s). Planck did not believe the quanta were real physical quantities. He considered them “a purely formal assumption” — a mathematical trick. He later admitted: "It was an act of desperation… I was ready to sacrifice any of my previous convictions about physics."
🤔 Why Didn't Planck Himself Believe in His Own Discovery?
Planck was by nature a conservative — a “reluctant revolutionary,” as historian Helge Kragh called him. Born on April 23, 1858, in Kiel, he grew up in an academic family. His professor Philipp von Jolly discouraged him from pursuing theoretical physics, telling him that “physics is a nearly completed science.”
Planck did not believe in atomism and did not accept the statistical interpretation of the second law of thermodynamics. Using Boltzmann's method was a last resort. Even afterwards, he spent years trying to bring quanta back within classical physics. Max Born wrote memorably: "He was, by nature, a conservative mind. He had nothing of the revolutionary. Yet his belief in the compelling force of logical reasoning from facts was so strong that he did not flinch from announcing the most revolutionary idea which ever has shaken physics."
Planck endured immense personal tragedies. His first wife, Marie Merck, died in 1909. His son Karl was killed in World War I in 1916. His twin daughters, Emma and Grete, died in 1917 and 1919 respectively. His son Erwin was executed by the Nazis in January 1945 for his role in the July 20 plot to assassinate Hitler.
🔬 How Did His Discovery Change Modern Physics?
Planck never proposed that light itself propagates as quanta. It was Albert Einstein who in 1905 took quanta seriously, using them to explain the photoelectric effect — that electrons are ejected from a metal surface only when the frequency of light exceeds a threshold, regardless of intensity. Einstein proposed that quanta are real particles — what we now know as photons.
Planck initially rejected the idea, saying “the theory of light would be thrown back centuries.” It was not until 1919 that he fully accepted that both emission and absorption of light are quantized.
The constant h proved that energy is not continuous but “granular” — a world-historic result. Niels Bohr used quanta in 1913 for the atomic model, Werner Heisenberg and Max Born in the 1920s laid the foundations of quantum mechanics, and today the Planck constant literally defines the Planck units — the physical limits of nature.
Planck was awarded the Nobel Prize in Physics 1918 "for the services he rendered to the advancement of physics by his discovery of energy quanta." He died on October 4, 1947, in Göttingen. The Max-Planck-Gesellschaft — 83 institutes across Germany — bears his name today.