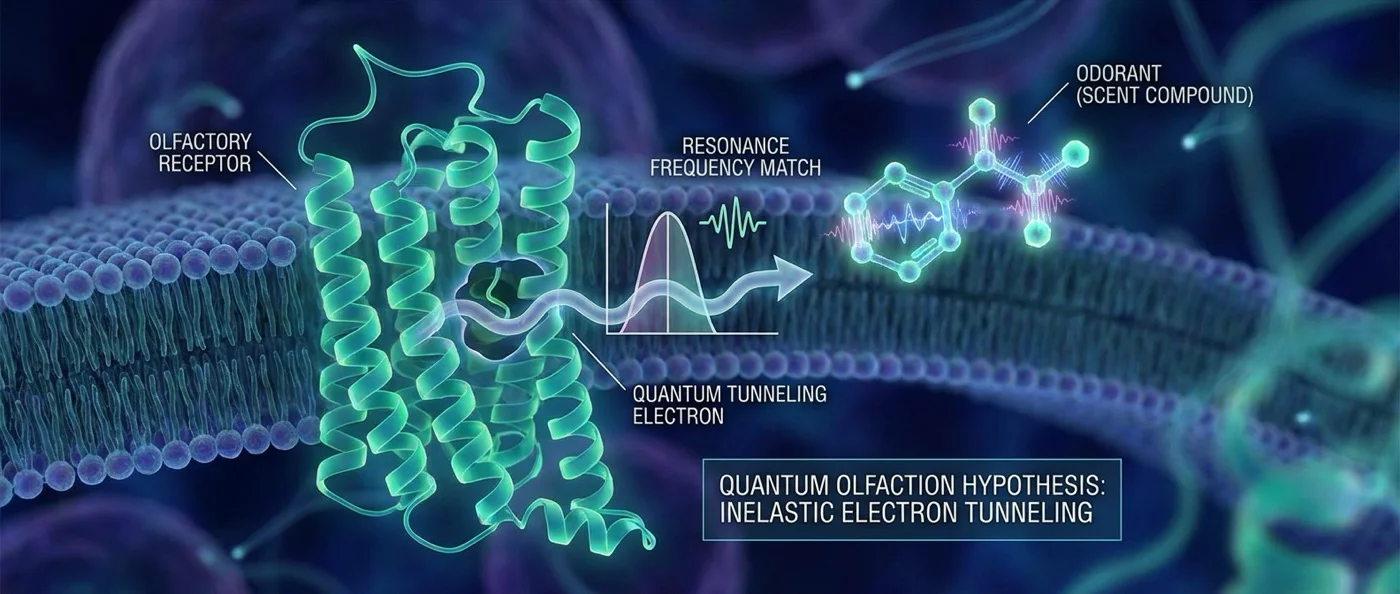
New theories propose that olfaction relies not only on molecular shape but on quantum vibrations. If true, quantum biology “sniffs” the world through electron tunneling.
📖 Read more: Quantum Photosynthesis: Plants' 100% Energy Efficiency
👃 A Mystery Hidden in Your Nose
Close your eyes and think of the scent of a freshly cut lemon. That sensation — so vivid, so immediate — seems simple. And yet, behind every breath hides one of biology's most enigmatic questions: how exactly does the nose recognize thousands of different molecules? The answer may lie in the most unexpected place — the world of quantum physics.
For decades, science accepted an elegant explanation: each odorant molecule fits into a receptor like a key in a lock. This model — known as the “shape theory” or docking theory — was based on the idea that a molecule's three-dimensional shape determined its smell. In 1991, Linda Buck and Richard Axel discovered olfactory receptors — a vast family of G-protein-coupled receptors — and won the Nobel Prize in Physiology in 2004. Humans possess approximately 400 functional olfactory receptor genes, while mice have over 1,000.
🔑 The Key That Doesn't Fit the Lock
If shape were the only criterion, molecules with the same form should always smell identical. But they don't. Benzaldehyde, for example, gives off an almond scent — but molecules with very different shapes and similar vibrational spectra smell alike. Even stranger: boranes, compounds containing no sulfur, smell like sulfur compounds. The B-H bond vibrates at a frequency similar to the S-H bond. Coincidence?
Malcolm Dyson didn't think so. As early as 1928, he proposed that a molecule's smell was due to its vibrational frequency in the infrared spectrum — not its shape. The idea was expanded by Robert H. Wright in 1954 but was soon abandoned in favor of shape theory. Until Luca Turin came along.
🧑🔬 Turin and the “Quantum Nose”
In 1996, the Italian-British biophysicist Luca Turin published a bold paper in the journal Chemical Senses. His proposal was radical: the G-protein-coupled receptors of the nose don't simply “see” the shape of molecules — they measure their molecular vibrations through inelastic quantum electron tunneling.
In Turin's “swipe card” model, the molecule must first fit into the receptor's binding site — like a card into a reader. But the “reading” happens by measuring vibrational energy: if the molecule's vibration matches the energy difference between two levels on the receptor, an electron passes through inelastic tunneling through the molecule, activating signal transduction.
The analogy with vision is no accident: just as retinal cone cells encode color through activity ratios at different light frequencies, olfactory receptors — according to Turin — encode smell through activity ratios at different vibrational frequencies. The nose, in a sense, “listens” to molecules.
🪰 Flies That Smell Quantum-Mechanically
The big moment came in 2007, when Marshall Stoneham and colleagues (University College London and Imperial College London) published in Physical Review Letters that Turin's mechanism was consistent with known physics — what they called “phonon-assisted tunneling.”
But the real test came from flies. In 2011, Franco, Turin, Mershin, and Skoulakis (MIT and the Alexander Fleming Biomedical Sciences Research Center) published a remarkable study in PNAS: Drosophila melanogaster flies could distinguish acetophenone from its deuterated version — molecules with exactly the same shape but different vibrations, due to replacing hydrogen with deuterium.
📖 Read more: Werner Heisenberg: The 24-Year-Old Who Discovered Matrix Mechanics
⚔️ The Counterattack: Human Receptors Say “No”
The story wasn't so simple. In 2004, Leslie Vosshall and Andreas Keller published in Nature Neuroscience that untrained volunteers could not distinguish acetophenone from its deuterated form. Turin accepted this result in 2013 but showed that for cyclopentadecanone — which has 28 hydrogens instead of 8 — volunteers could recognize the difference. More C-H bonds meant a stronger “vibrational signal.”
But in 2015 came the hardest challenge. Eric Block and colleagues published in PNAS under the title “Implausibility of the Vibrational Theory of Olfaction.” Studying the human musk receptor OR5AN1, they found it responded identically to normal and deuterated versions of cyclopentadecanone and muscone. Additionally, the mouse thiol receptor MOR244-3 reacted the same to normal, deuterated, and carbon-13 isotopologues.
Vosshall commented: “The data do not support the vibration theory.” Richard Axel, co-recipient of the 2004 Nobel Prize for olfaction, emphasized that only a “microscopic look at the receptors of the nose will finally show what is at work.”
🧪 The Carvone Story: An Enigma Within an Enigma
Perhaps the most fascinating point in this debate is carvone. This molecule exists in two enantiomers — right-handed and left-handed. They have exactly the same vibrations. Yet one smells like mint and the other like caraway. If smell depended only on vibrations, this shouldn't happen.
Turin responded with an elegant experiment, filmed for BBC Horizon: by mixing 60% butanone with 40% mint-carvone, trained perfumers identified the smell as caraway. The explanation: the carbonyl group of mint-carvone was blocked by the receptor, while butanone “filled in” the vibrational information. Shape and vibration work together.
🧬 Quantum Biology: The Nose Isn't Alone
Quantum smell is not an isolated phenomenon. It belongs to the broader field of quantum biology, where quantum phenomena influence biological processes at room temperature. Photosynthesis exploits quantum coherence to transfer energy with nearly 100% efficiency. Migratory birds use radical pairs in the cryptochrome protein of their retinas to sense the Earth's magnetic field — a phenomenon based on quantum entanglement. And enzymes appear to use quantum proton tunneling in biochemical reactions.
In this context, the idea that the nose operates as a “quantum spectrometer” isn't outlandish — it's simply controversial.
⚖️ Two Theories, One Nose
Today, the scientific community has not reached a definitive conclusion. Shape theory explains many things but not all. Vibration theory explains some anomalies but faces serious experimental objections. More and more researchers believe the reality lies somewhere in between — a hybrid model where the molecule must first fit (shape), but the final recognition also depends on vibrations within the receptor.
In the end, what makes this debate so fascinating isn't just the answer — it's the question. Quantum physics, born to explain atoms and electrons, may be hiding inside our most ancient and primal sense: smell. Every time you smell a rose, you might not just be “touching” a molecule — you might be “listening” to its quantum songs.