Einstein won the 1921 Nobel Prize in Physics not for relativity, but for explaining the photoelectric effect — proving the particle nature of light.
📖 Read more: Quantum Physics: Does God Play Dice?
✨ The spark that started it all
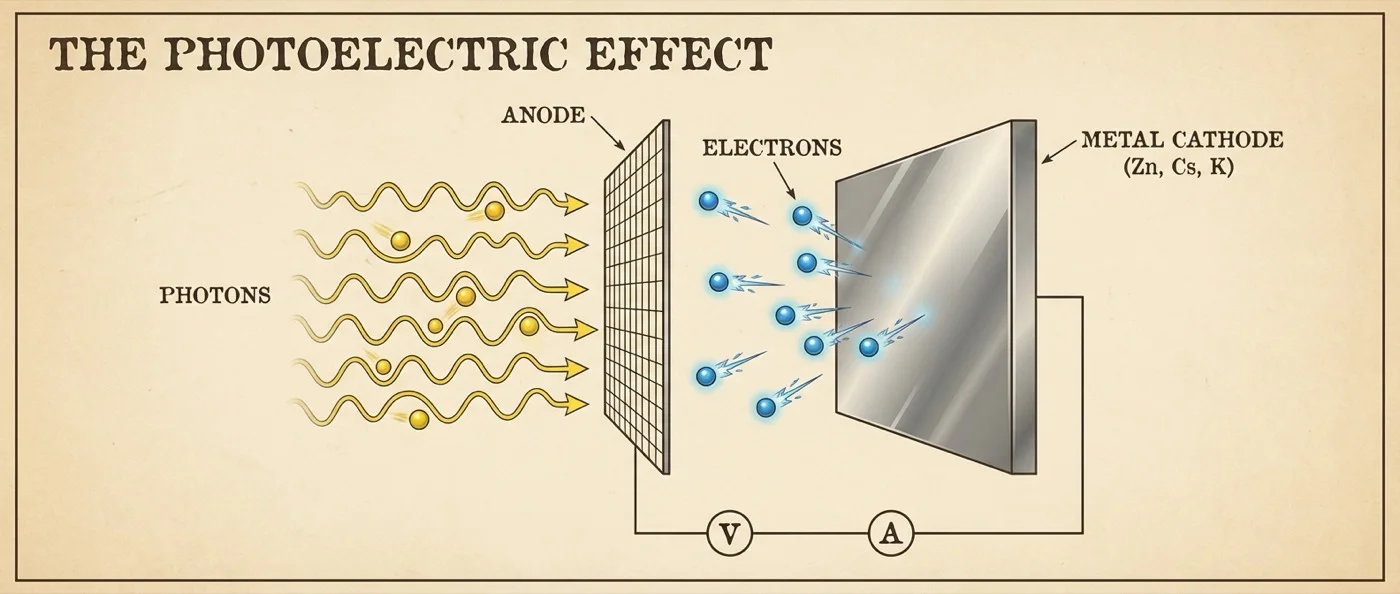
The story begins in a darkened laboratory in Karlsruhe, 1887. Heinrich Hertz — the physicist who had just experimentally proven the existence of electromagnetic waves — noticed something unexpected. While studying sparks between metal electrodes, he observed that the sparks grew stronger when ultraviolet light struck the electrodes. The phenomenon was clear, yet inexplicable: why did light exactly help the sparks?
Hertz recorded the phenomenon in his publication but never lived to explain it — he died at just 36. The baton passed to Wilhelm Hallwachs, who in 1888 discovered that a freshly cut zinc plate, exposed to ultraviolet light, emits negatively charged particles. It charged positively with no other external cause. A new world was opening up.
🔬 Lenard finds the impossible results
The most critical turning point came between 1899 and 1902, in the laboratory of Philipp Lenard. Lenard — a former assistant of Hertz — carried out a series of elegant experiments. Enclosing metal electrodes in evacuated glass tubes and illuminating them with UV light of varying intensities, he measured something that fit nowhere in classical physics.
According to Maxwell's wave theory, the energy of emitted electrons should increase with the intensity of light — brighter light, faster electrons. Instead, Lenard found that intensity did not change the electron energy at all. Only the frequency of the light mattered. Low frequency, no electrons — regardless of how bright the light. High frequency, electrons immediately — even if the beam was faint.
"The energy of emitted electrons was independent of light intensity — a result entirely incompatible with wave theory."
— Philipp Lenard, Annalen der Physik, 1902The classical model also predicted that electrons would “gather” energy gradually from the light wave — meaning there would be a measurable time delay. Lenard found emission in less than a nanosecond. Instantaneous. It was as if the energy struck the electron all at once.
💡 A patent clerk changes everything
March 1905. A 26-year-old clerk at the Swiss Patent Office in Bern, living in a small apartment with his wife Mileva and their son Hans Albert, sits down to write. Albert Einstein belonged to no university. He had no laboratory, no funding, no access to experimental data except through scientific journals.
At home, with paper and pencil, he synthesized an idea so radical that he himself called it “heuristic” — a proposal without full proof, but with enormous explanatory power. His paper, "Über einen die Erzeugung und Verwandlung des Lichtes betreffenden heuristischen Gesichtspunkt" (On a Heuristic Viewpoint Concerning the Production and Transformation of Light), arrived at Annalen der Physik on 18 March and was published on 9 June 1905.
His proposal was simple and sweeping: light is not a continuous wave, but consists of “packets” of energy — quanta. Each quantum carries energy equal to E = hν, where h is Planck's constant and ν the frequency. A light quantum — later named photon by Gilbert N. Lewis in 1926 — carries energy in packets, like a bullet rather than a wave. If a photon's energy exceeds the work function W of the metal (the minimum binding energy of an electron), the electron is emitted with kinetic energy:
Kmax = hν − W
The equation explained everything: why there is a threshold frequency (ν₀ = W/h), why intensity doesn't change the energy (each photon acts individually), why emission is instantaneous (one photon strikes one electron). Lenard's mysterious world suddenly made sense.
📖 Read more: Why Quantum Physics & Relativity Can't Be Unified
⚠️ Opposition that lasted a decade
The reaction was near-universal rejection. Even Max Planck — the very man who first introduced the energy quantum for black-body radiation in 1900 — considered light quanta too bold. Planck accepted that emission of energy occurs in quanta, but insisted that light itself remained a wave. Niels Bohr was equally skeptical. Quantizing light seemed to demolish Maxwell's equations — a theory tested and supremely powerful.
But the toughest critic came from Southern California. Robert A. Millikan, an experimental physicist at the University of Chicago, considered the particle theory of light “quite unthinkable” and set out to disprove it experimentally. He spent nearly a decade (1905–1916) on exquisitely precise experiments, designing unique apparatus to accurately measure the relationship between light frequency and electron kinetic energy.
In 1914, after years of measurements, Millikan announced the result: Einstein's equation was exactly correct. The energy-frequency graph was a perfectly straight line, with slope equal precisely to Planck's constant h. Millikan determined h with unprecedented accuracy. Yet, in one of the most ironic moments in physics, he declared that while his results confirmed Einstein's equation, the “corpuscular theory” of light remained for him “unthinkable.”
🏅 A Nobel no one expected
In November 1922, the Swedish Academy announced that the 1921 Nobel Prize in Physics — which had gone unawarded the previous year because no nominee met the criteria — was bestowed upon Albert Einstein "for his services to Theoretical Physics, and especially for his discovery of the law of the photoelectric effect." Not for relativity — general relativity was still disputed in some circles. The award's wording even betrayed some hesitation: it did not explicitly acknowledge the particle nature of light.
On 10 December 1922, Einstein was not in Stockholm — he was traveling in Japan. The German ambassador accepted the prize in his place. Einstein's Nobel lecture, delivered in July 1923 in Gothenburg, was not even about the photoelectric effect — he spoke about the theory of relativity.
That same year, 1923, Millikan too received the Nobel “for his work on the elementary electric charge and on the photoelectric effect” — that is, for experiments originally designed to disprove Einstein.
🌌 An idea that changed physics
Full acceptance of the particle nature of light came only after 1924, when Satyendra Nath Bose mathematically derived the Planck spectrum using particle statistics. The so-called Bose-Einstein statistics provided the theoretical confirmation: photons were not merely a “heuristic idea” but real entities. From there, quantum mechanics unfolded rapidly — Einstein's 1905 insight proved to be the seed of an entire scientific revolution.
Today, the equation Kmax = hν − W is taught in every introductory quantum course — so simple, so fundamental. The photoelectric sensor is everywhere: in digital cameras, photovoltaic panels, night-vision devices, and particle detectors. But beyond the applications, the story of the photoelectric effect remains a lesson about the nature of discovery: sometimes, the deepest truth lies in the result that doesn't fit.