Every year, drug-resistant bacteria kill over 1.2 million people worldwide — and the toll is rising. Antibiotics, once the crowning achievement of modern medicine, are steadily losing their edge. A team of biologists at the University of California, San Diego has just unveiled a tool that doesn't target the bacteria themselves — it targets the very genes that make them resistant.
📖 Read more: RNA Nanostars Create Artificial Compartments Inside Bacteria
🧬 What Is Antibiotic Resistance and Why Should We Worry
Antimicrobial resistance (AMR) isn't a future threat. It's already here. Bacteria that were once easily treated with penicillin or amoxicillin now require “last-resort” antibiotics — and even those are beginning to fail. According to current estimates, if nothing changes dramatically, resistant “superbugs” will claim over 10 million lives annually by 2050.
Bacteria develop resistance through several mechanisms. The most alarming involves plasmids — circular DNA segments that replicate within cells and carry resistance genes from one bacterium to another. This means resistance spreads even without inheritance — like a contagious disease among microbes.
🔬 The New Technology: pPro-MobV — A Gene Drive for Bacteria
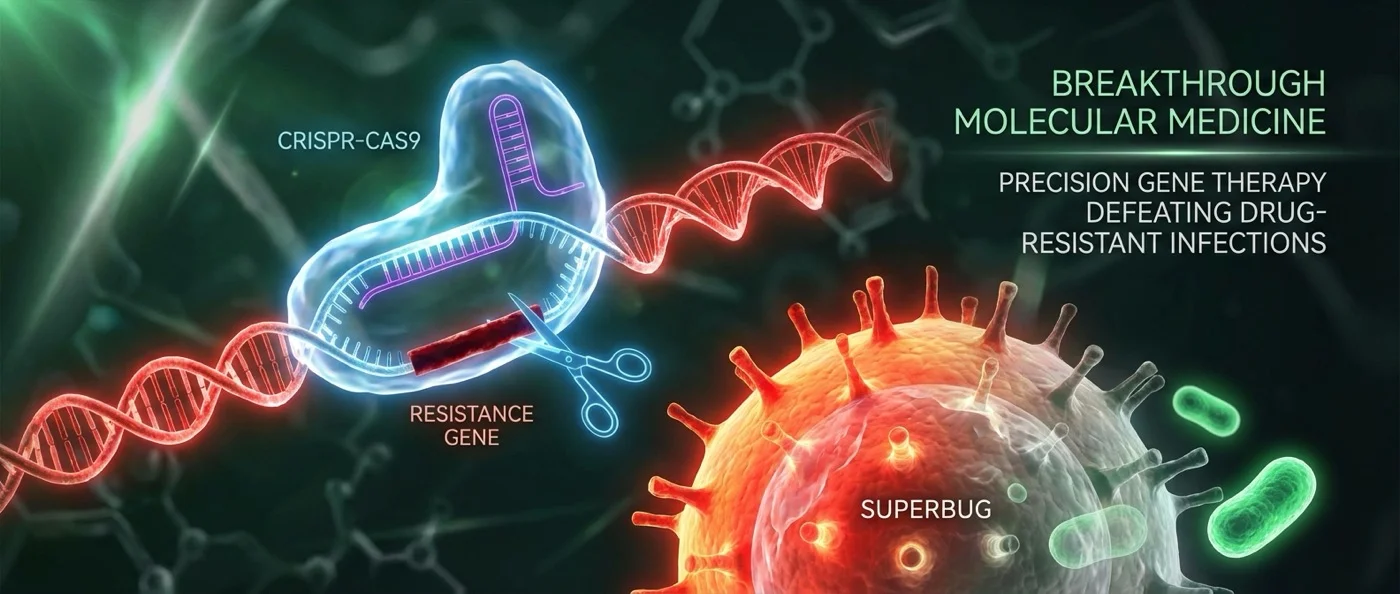
The team led by Professors Ethan Bier and Justin Meyer at UC San Diego developed a CRISPR-based system that works like a “gene drive” — but instead of targeting insects, it targets bacteria. The tool, called pPro-MobV, is a second-generation version of the Pro-Active Genetics (Pro-AG) technology originally developed in 2019 in collaboration with Professor Victor Nizet.
The core principle: a CRISPR genetic “cassette” is introduced into a few bacteria. There, the cassette precisely cuts the antibiotic resistance genes located on plasmids. But the real breakthrough comes next — the cassette then copies itself and spreads to neighboring bacteria through conjugal transfer, a form of bacterial “mating.”
"With pPro-MobV we have brought gene-drive thinking from insects to bacteria as a population engineering tool. With this new CRISPR-based technology we can take a few cells and let them go to neutralize antibiotic resistance in a large target population."
— Ethan Bier, Professor of Cell and Developmental Biology, UC San Diego🦠 How It Works: Spreading From Cell to Cell
The first-generation Pro-AG system (2019) could introduce the genetic cassette into bacteria, but its spread was limited. The new pPro-MobV exploits a phenomenon called the “conjugal tunnel” — a natural channel bacteria create to exchange genetic material. Through this tunnel, the CRISPR elements automatically transfer from a donor cell to its neighbors, creating a chain reaction.
The team demonstrated that the system works even inside biofilms — colonies of microorganisms that form a protective layer on surfaces. Biofilms are a clinical nightmare: antibiotics struggle to penetrate them, and the majority of serious hospital-acquired infections are biofilm-related. The ability of pPro-MobV to operate within these communities is critical.
💡 The Difference From Conventional Antibiotics
Traditional antibiotics kill bacteria — but create evolutionary pressure that favors the most resistant survivors. pPro-MobV doesn't kill a single bacterium. Instead, it strips them of their “weapons” — resistance genes — so that conventional antibiotics become effective once again.
📖 Read more: Chimpanzees Drink Alcohol in Wild — And Keep Coming Back
🧪 Bacteriophages as Allies
The researchers went a step further: they discovered that pPro-MobV components can also be delivered via bacteriophages — viruses that exclusively infect bacteria. Bacteriophages (or “phages”) are natural enemies of bacteria and are already being intensively studied as an alternative therapy in the post-antibiotic era. The idea is to combine engineered phage delivery with pPro-MobV, creating a powerful “duo” that infiltrates bacteria, bypasses their defenses, and disables resistance genes.
🏥 Where It Could Be Applied
The technology's applications extend far beyond hospitals. Antibiotic resistance thrives in healthcare settings, sewage treatment facilities, fish farms, and animal husbandry operations. In these environments, bacteria form dense biofilms on pipes, tanks, and equipment — an ideal theater of operations for this new technology.
"If you could reduce the spread from animals to humans you could have a significant impact on the antibiotic resistance problem since roughly half of it is estimated to come from the environment."
— Ethan Bier, UC San Diego🔒 Built-In Safety Mechanism
A reasonable concern: what if the genetic cassette spreads uncontrollably? The team built in a safety mechanism based on “homology-based deletion.” This technique allows the CRISPR cassette to be removed if deemed necessary, functioning as a “kill switch” for the entire system.
"This technology is one of the few ways that I'm aware of that can actively reverse the spread of antibiotic-resistant genes, rather than just slowing or coping with their spread."
— Justin Meyer, Professor of Ecology, Behavior and Evolution, UC San Diego⚡ What This Means for the Future
The publication in npj Antimicrobials and Resistance (a Nature journal) marks a significant milestone. However, this is still laboratory research — clinical application will require many more years of testing. Nevertheless, the working principle of pPro-MobV changes the game: instead of constantly searching for new antibiotics in a never-ending arms race with bacteria, we can disarm resistance itself.
The technology opens paths in three directions: clinical medicine (combating hospital-acquired infections), environmental remediation (cleaning contaminated water and soil), and microbiome engineering (reprogramming bacterial populations). If clinical results confirm the laboratory promises, this could represent a paradigm shift in addressing one of the greatest health crises of the 21st century.
Sources: